Skin Acids

<image, oxidation of razor blade,istockphoto.jpg>
Propionic Acid
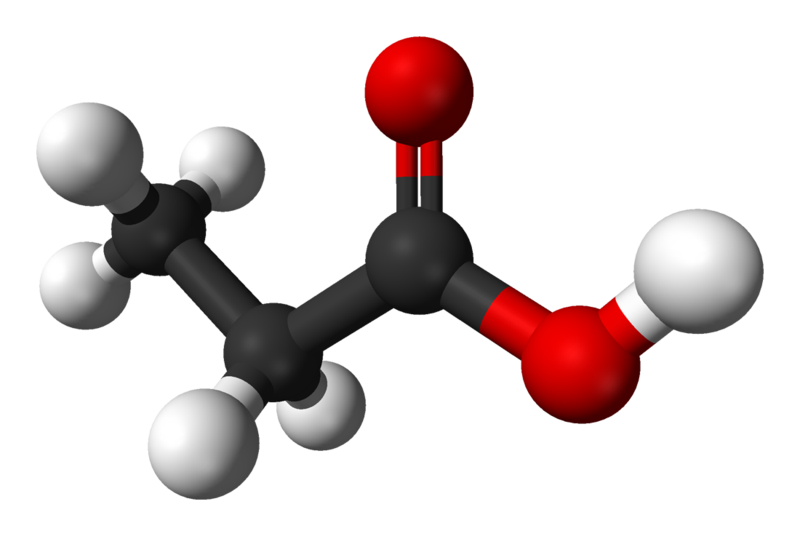
<image, propionic acid, 800px-Propionic-acid-3D-balls.png>
Propionic acid was first described in 1844 by Johann Gottlieb, who found it among the degradation products of sugar. Over the next few years, other chemists produced propionic acid in various other ways, none of them realizing they were producing the same substance. In 1847, the French chemist Jean-Baptiste Dumas established that all the acids were the same compound, which he called propionic acid, from the Greek words protos = "first" and pion = "fat," because it was the smallest H(CH2)nCOOH acid that exhibited the properties of the other fatty acids, such as producing an oily layer when salted out of water and having a soapy potassium salt.
Propionic acid inhibits the growth of mold and some bacteria. As a result, most propionic acid produced is used as a preservative for both animal feed and food for human consumption, and can be used as a preservative for Ballistics Gel. For animal feed, it is used either directly or as its ammonium salt. In human foods, especially bread and other baked goods, it is used as its sodium or calcium salt. Similar usage occurs in some of the older anti-fungal foot powders.
Propionic acid is also useful as a chemical intermediate. It can be used to modify synthetic cellulose fibers. It is also used to make pesticides and pharmaceuticals. The esters of propionic acid are sometimes used as solvents or artificial flavorings.
The human skin is host to a species of bacteria known as Propionibacterium acnes, which is named after its ability to produce propionic acid. This bacteria lives mainly in the sebaceous glands of the skin and is one of the principle causes of acne.
http://en.wikipedia.org/wiki/Propionic_acid
Acid Properties
The word acid comes from the Latin word acere, which means "sour." All acids taste sour. Well known from ancient times were vinegar, sour milk and lemon juice. Aspirin tastes sour if you don't swallow it fast enought. Its scientific name is acetosallicylic acid! Other languages derive their word for acid from the meaning of sour. So, in France, we have acide. In Germany, we have säure from saure and in Russia, kislota from kisly.
In 1663, Robert Boyle wrote that acids would make a blue vegetable dye called "litmus" turn red.
Acids destroy the chemical properties of bases
Acids conduct an electric current

<image, Acid dissolves a metal releasing Hydrogen Gas, explodeCan-w.jpg>
Upon chemically reacting with an active metal, acids will evolve hydrogen gas (H2). The key word, of course, is active. Some metals, like gold, silver or platnium, are rather unreactive and it takes rather extreme conditions to get these "unreactive" metals to react. Not so with the metals in this property. The include the alkali metals (Group I, Li to Rb), the alkaline earth metals (Group II, Be to Ra), as well as zinc and aluminum. Just bring the acid and the metal together at anything close to room temperature and you get a reaction. Here's a sample reaction:
Zn + 2 HCl(aq) ---> ZnCl2 + H2
Another common acid reaction some sources mention is that acids react with carbonates (and bicarbonates) to give carbon dioxide gas:
HCl + NaCO3 ---> CO2 + H2O + NaCl
Acid-Base-Properties.html

<image, HCl and NaOH, Bottle-HCl&NaOH.JPG>

<litmus paper, 75020699.JPG>
Chemical Effects:
You may be wondering what acids and bases do to other elements. Acids dissolve metals – the acid in tomato sauce dissolves an aluminum pan, acids break up proteins –vinegar in marinating meat, acids conduct electricity - battery acid and dissolve carbonate compounds - bacterial acids dissolve enamel forming cavities on teeth. Bases and acids neutralize each other. Mixing equal amounts of similar strength acids and bases produces a neutral substance.
actacidbase.html
Acids are exceptionally reactive compounds which we find in our every day lives. They usually share the following properties.
A characteristic sour taste;
ability to change the color of litmus paper from blue to red;
react with certain metals to produce gaseous H2 ;
react with bases to form a salt and water.
Common household acids include vinegar, lemon juice and automobile battery acid. In their weaker forms they can add a sour taste to our food; in their stronger forms they are corrosive agents which can burn skin, and be used to etch or dissolve metal and even glass.
Examples of acids found in the either the laboratory or the work place include:
Hydrochloric Acid: Also known as Muriatic Acid, this is a highly corrosive acid and is often used to clean calcium carbonate build up from the inside of kettles or from around water faucets and from shower heads;
Sulphuric Acid: This is a common acid in both the laboratory and industry. It is both highly corrosive and economical to manufacture, which makes it the reagent of choice for many applications;
Phosphoric Acid: This acid is used to remove rust and rust stains from metal tools and from car bodies undergoing repairs;
Nitric Acid: This is another common laboratory acid used as a reagent in many chemical tests and experiments due to the fact that almost all of its products (salts) are soluble in water;
Hydrofluoric Acid: This acid is extremely corrosive and has the unique property of being able to etch (eat away) glass. Consequently it is used in industry to write signs on glass windows in stores and office buidings or on glass products.
The key to understanding acids is to note that acids and solutions of their salts all conduct electricity. This implies the existence of charged particles that migrate under the influence of an electric field. Acids are acids by virtue of the presence of an excess of hydrogen ions in the solution, Their salts are created when the positive hydrogen ions are replaced with positive metal ions, for example when HCL reacts with Sodium (Na) to produce NaCl with the release of H2 gas.
10081/

<image, nitric acid, silver-recovery-NO2-a.jpg>
Corrosive Nature
Many substances, such as ionic compounds, dissolve in acid no differently than they dissolve in water. However, metals can also be dissolved in acids, but this is by an entirely different mechanism, and it's not really dissolving at all. When some metals are added to acids, a reaction takes place which oxidizes the metal, or removes electrons from the atoms. This makes metal ions from the neutral atoms, and these ions dissolve. At the same time hydrogen gas (H2) is created with these electrons that were pulled from the metal. This is very different that just dissolving salt in water, where no chemical reaction has taken place at all. Once you add a metal to acid, it is not simple to return to the metal. If you add salt to water and dissolve it, you can just let the water dry up and the salt will return just as it was before.

<steel and acid, SeanHealy_Nov06c.jpg>
Human Skin
So what is skin?
Skin is a miracle garment. It's soft, pliable, strong, waterproof, and self-repairing.
What would you be like without skin?
The answer is, quite simply, a big squishy mess! Your skin is like a very large container. It's the largest organ of your body, and without it, all your delicate insides would spill right out.
Skin doesn't just cover you!
Your skin doesn't just cover you. It does a whole lot more. It functions as protective wrapping. Along with a layer of fat underneath, it insulates you against all kinds of bumps, bangs and wear and tear. It keeps germs and water OUT (unless you have a break in your skin) and keeps your body's fluids and salts IN.
Skin manufactures and oozes out all sorts of wonderful liquids. Waxes and oils act as your body's natural waterproofer and a protector against germs. They make your skin softer; but they can also give you pimples. Your skin also contains glands which manufacture sweat. With sweat, not only does your body get cooled by its evaporation, but it has a convenient way to get rid of chemicals it doesn't need.
How does it do all this?
Skin is alive. It's made of many thin sheets of layers of flat, stacked cells in which you'll find nerves, blood vessels, hair follicles, glands, and sensory receptors.
Older cells are constantly being pushed to the surface by new cells which grow from below. When the old ones reach the top, they become wider and flatter as they get rubbed and worn by all your activity. And, sooner or later, they end up popping off like tiles blown from a roof in a strong wind. In fact, every minute 30,000-40,000 dead skin cells fall from your body! In approximately a month's time, your body has made a whole new layer of skin cells!
Ever wonder what makes different skin colors?
A pigment called melanin. More melanin in your skin cells makes your skin darker, less makes it lighter. Sitting in the sun can also cause more melanin to be manufactured in your skin cells. The result? A suntan.
FACTOIDS:
As an adult, you may have more than 20 square feet of skin -- about the size of a blanket.
You are likely to shed some 40 pounds of skin in a lifetime.
Right now there are over a million dust mites, microscopic critters invisible to the naked eye, on your mattress and pillow, chomping on the dead skin cells that fell off you last night!
pg000146.html
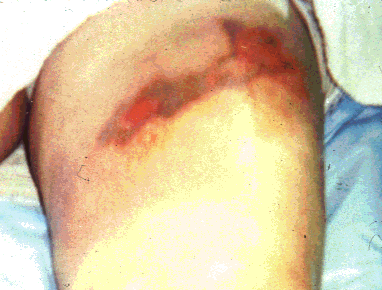
<acid burns, hfburn.gif>
Human skin is a remarkable organ, the body's largest, but it is often taken for granted. Most people are content to let skin be until dryness, oiliness, a rash or a wrinkle rouses attention. But once they understand how skin functions, many reconsider the importance of the skin and the quality and content of the skin care products they use. Using natural skin care can make more of a difference than most folks realize.
Consider these facts:
1 . An adult's skin comprises between 15 and 20 percent of the total body weight.
2. Each square centimeter has 6 million cells, 5,000 sensory points, 100 sweat glands and 15 sebaceous glands.
Skin is constantly being regenerated. A cell is born in the lower layer of the skin called the dermis, which is supplied with blood vessels and nerve ending. The cell migrates upward for about two weeks until it reaches the bottom portion of the epidermis, which is the outermost skin layer. The epidermis doesn't have blood vessels but does have nerve endings. The cell spends another two weeks in the epidermis, gradually flattening out and continuing to move toward the surface. Then it dies and is shed.
Two billion to 3 billion skin cells are shed daily. The body expends this effort to replace skin every month because the skin constitutes the first line of defense against dehydration, infection, injuries and temperature extremes. Skin cells can detoxify harmful substances with many of the same enzymatic processes the liver uses. The unbroken surface also prevents infectious organisms from penetrating into systemic circulation. As gatekeeper, the skin absorbs and uses nutrients applied topically. Because it cannot completely discriminate, the skin may absorb the synthetic chemicals often present in soaps and lotions and other skin care products, which at best it has no use for and at worst can be toxic or irritating.
Most of our site visitors are committed to natural foods and remedies, but many aren't as selective when it comes to personal skin care products. These otherwise savvy shoppers might purchase any sale shampoo, skin care cleanser or lotion. But because new skin is constantly being generated and because it plays such an important protective role, it makes sense to choose nourishing natural skin care products.
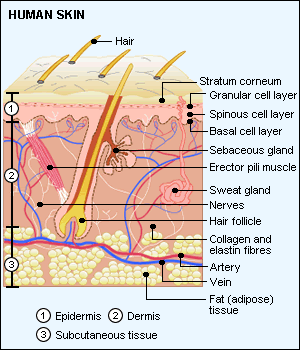
<skin diagram, skinstructure.gif>
The Epidermis
The epidermis is the topmost layer of the skin. It is the first barrier between you and the outside world. The epidermis consists of three types of cells keratinocytes, melanocytes and langerhans cells. Keratinocytes, the cells that make the protein keratin, are the predominant type of cells in the epidermis. The total thickness of the epidermis is usually about 0.5 - 1 mm. At the lowermost portion of the epidermis are immature, rapidly dividing keratinocytes. As they mature, keratinocytes lose water, flatten out and move upward. Eventually, at the end of their life cycle, they reach the uppermost layer of the epidermis called stratum corneum. Stratum corneum consists mainly of dead keratinocytes, hardened proteins (keratins) and lipids, forming a protective crust. Dead cells from stratum corneum continuously slough off and are replaced by new ones coming from below. The skin completely renews itself every 3 - 5 weeks. Most mild peels work by partly removing the stratum corneum and thus speeding up skin renewal.
Another significant group of cell in the epidermis are melanocytes, the cells producing melanin, the pigment responsible for skin tone and color. Finally, Langerhans cells are essentially the front door of the immune system in the epidermis. They prevent unwanted foreign substances from penetrating the skin.
The condition of epidermis determines how "fresh" your skin looks and also how well your skin absorbs and holds moisture. Wrinkles, however, are formed in lower layers.
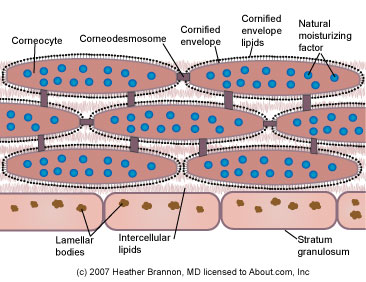
<stratum corneum, Stratum-Corneum.jpg>
The epidermis consists of many layers:
The stratum corneum, or outer layer: This layer is made of flattened epithelial cells in multiple layers. These layers are called keratinized layers because of the buildup of the protein keratin in those cells. Keratin is a strong protein that is specific to the skin, hair and nails. This layer of skin is, for the most part, dead. It is composed of cells that are almost pure protein.
The translucent or transitional layer: This is a translucent, thin layer of cells. This layer is sometimes visible in thick skin; however, nuclei and other organelles are not visible. The cytoplasm (the amorphous area between the nucleus and the outer membrane of the cell) is mostly made of keratin filaments.
The suprabasal layers. This is three to five layers of flattened polygonal cells that have granules in the cytoplasm. Below them is a layer of cube-shaped cells that also contain bundles of keratin filaments.
The basal or cell-division layer: This layer is just above the basement membrane and the dermis. It is a single layer of cells that undergo cell division to renew the upper layers of the epidermis.
Since the human epidermis is renewed every 15-60 days, proper surface nutrition feeds the cells of the basal layer. Exfoliation will remove dry or damaged skin of the outer layer to allow newer cells to be visible.
The Dermis
The dermis is the middle layer of the skin located between the epidermis and subcutaneous tissue. It is the thickest of the skin layers and comprises a tight, sturdy mesh of collagen and elastin fibers. Both collagen and elastin are critically important skin proteins: collagen is responsible for the structural support and elastin for the resilience of the skin. The key type of cells in the dermis is fibroblasts, which synthesize collagen, elastin and other structural molecules. The proper function of fibroblasts is highly important for overall skin health.
The dermis also contains capillaries (tiny blood vessels) and lymph nodes (depots of immune cells). The former are important for oxygenating and nourishing the skin, and the latter -- for protecting it from invading microorganisms.
Finally, the dermis contains sebaceous glands, sweat glands, hair follicles as well as a relatively small number of nerve and muscle cells. Sebaceous glands, located around hair follicles, are of particular importance for skin health as they produce sebum, an oily protective substance that lubricates and waterproofs the skin and hair. When sebaceous gland produce too little sebum, as is common in older people, the skin becomes excessively dry and more prone to wrinkling. Conversely, overproduction or improper composition of sebum, as is common in adolescents, often leads to acne.
The dermis is the layer responsible for the skin's structural integrity, elasticity and resilience. Wrinkles arise and develop in the dermis. Therefore, an anti-wrinkle treatment has a chance to succeed only if it can reach as deep as the dermis. Typical collagen and elastin creams, for example, never reach the dermis because collagen and elastin molecules are too large to penetrate the epidermis. Hence, contrary to what some manufacturers of such creams might imply, these creams have little effect on skin wrinkles.
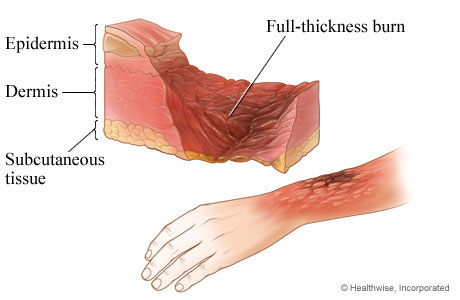
<burn and subcutaneous tissue, hwkb17_003.jpg>
Subcutaneous tissue
Subcutaneous tissue is the innermost layer of the skin located under the dermis and consisting mainly of fat. The predominant type of cells in the subcutaneous tissue is fat cells. Subcutaneous fat acts as a shock absorber and heat insulator, protecting underlying tissues from cold and mechanical trauma. Interestingly, most mammals lack subcutaneous tissue because their fur serves as a shock absorber and heat insulator. Sweat glands and minute muscles attached to hair follicles originate in subcutaneous tissue.
The loss of subcutaneous tissue, often occurs with age, leads to facial sag and accentuates wrinkles. A common procedure performed by dermatologists to counteract this process is to inject fat (collected elsewhere in the body) under the wrinkles on the face.
natbeauty.asp
Microorganisms in Skin
The surface of the skin itself comprises several distinct environments. Areas such as the axilla (armpit), the perineum (groin) and the toe webs provide typically moister regions for bacterial growth. These "tropical forest" environments often harbor the largest diversity amongst the skin flora. Typical organisms include Staphylococcus aureus, Corynebacterium and some Gram-negative bacteria. The bulk of the human skin surface, however, is much drier and is predominantly inhabited by Staphylococcus epidermidis and Propionobacterium.
normal.htm
<image, skin bacteria, 199812-026-Staph-Bacteria.jpg>
Several groups of microorganisms cause skin infections: bacteria, fungi (molds and yeast), and viruses. Based on the appearance of the infection, the physician determines what group of microorganisms is likely causing the infection, then he or she collects a specimen for one or more types of cultures. A sample of material--such as skin cells, pus, or fluid--is taken from the infection site, placed in a sterile container, and sent to the laboratory. In the laboratory, each type of culture is handled differently.
Bacterial infections are the most common. Bacteria cause lesions, ulcers, cellulitis, and boils. Pyoderma are pus-containing skin infections, such as impetigo, caused by Staphylococcus or group A Streptococcus bacteria. To culture bacteria, a portion of material from the infection site is spread over the surface of a culture plate and placed in an incubator at body temperature for one to two days. Bacteria in the skin sample multiply and appear on the plates as visible colonies. They are identified by noting the appearance of their colonies, and by performing biochemical tests and a Gram's stain.
The Gram's stain is done by smearing part of a colony onto a microscope slide. After it dries, the slide is colored with purple and red stains, then examined under a microscope. The color of stain picked up and retained by the bacteria (purple or red), their shape (such as round or rectangle), and their size provide valuable clues as to their identity.
A sensitivity test, also called antibiotic susceptibility test, is also done. The bacteria are tested against different antibiotics to determine which will effectively treat the infection by killing the bacteria.
Fungal cultures are done less frequently. A group of fungi called dermatophytes cause a skin infection called ringworm. Yeast causes an infection called thrush. These infections are usually diagnosed using a method other than culture, such as the KOH test. A culture is done only when specific identification of the mold or yeast is necessary. The specimen is spread on a culture plate designed to grow fungi, then incubated. Several different biochemical tests and stains are used to identify molds and yeasts.
Viruses, such as herpes, can also cause skin infections. Specimens for viral cultures are mixed with commercially-prepared animal cells in a test tube. Characteristic changes to the cells caused by the growing virus help identify the virus.
Results for bacterial cultures are usually available in one to three days. Cultures for fungi and viruses may take longer--up to three weeks. Cultures are covered by insurance.
skin_culture.jsp
<image, bacteria on skin, fig027.jpg>
Hygiene
To cut down on the number of bacteria on the human body, good hygiene should be practiced. The following research shows the effectiveness of Razor-Gator in the reduction of bacteria present on the razor blade.
Plate 1: head of razor swabbed immediately after shave and water rinse.

|
Plate 1: head of razor swabbed immediately after shave and water rinse.
Results: Razor Gunk contains 'billions' of bacterial colonies, as seen by the growth on the agar plates. |

|
Plate 2. head of razor swabbed 24 hours after shave and water rinse.
Results: Razor Gunk supports the growth of bacteria even when the blade is left to dry for 24 hours. Material plated on agar grew as many colonies as did plate 1. |
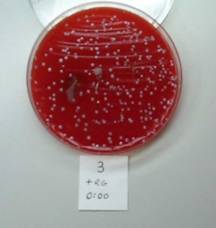
|
Plate 3. head of razor swabbed immediately after shave, and use of RAZOR GATOR
Results: Razor Gator cleans and sanitizes the blade head, removing over 99% of the bacteria present in razor gunk. |
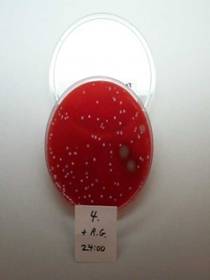 |
Plate 4. head of razor swabbed 24 hours after shave and use of RAZOR GATOR.
Results: Razor Gator removes over 99% of the bacterial colonies present, thus reducing their subsequent growth over 24 hours. |
http://razor-gator.com/razor_gator_research.htm